By Natalie Pape, MA RDN
Recently, interest in collagen has exploded in both the athletic and beauty worlds. Supplement companies are catering to this increasing demand: the global collagen peptides market is expected to reach $6.8 billion by 2025. Perusing the shelves at your local drug or grocery store, you will find a variety of collagen pills, powders, and gummy supplements. Even food companies are cashing in by adding collagen to baked goods, protein bars, oatmeal, even coffees, and teas- all promising to support hair, skin, nail, and joint health.
This nutrition topic comes at a time when I, personally, have started to feel the first signs of joint pain from lifting (stupid knees!) as well as the first signs of fine lines forming on my face. Hello, 30s! Is there substance to the collagen hype? Will taking supplemental collagen help with my bum knees or restore the youth of my 20s?
First, what even is collagen?
What Is Collagen?
Collagen is the main and most abundant structural protein in the human body, making up about 25 to 30 percent of total protein body mass. Collagen is considered a fibrous protein: These molecules are the major structural material of animal tissue. Specifically, collagen provides strength and structure to connective tissues in bones, skin, tendons, ligaments, the gut, cartilage, scar tissue, and blood-vessel walls. Its main purpose is to hold these tissues together and help them withstand stretching. The etymology of the word itself has roots in the Greek kolla which translates to “glue”- possibly referring to collagen’s structural role in connective tissue; it’s the “glue” of life. It could also be a reference to the process of boiling collagen-rich animal parts to form glue. As a vegan with a clear bias toward showing kindness to animals, I am going to side with the former.
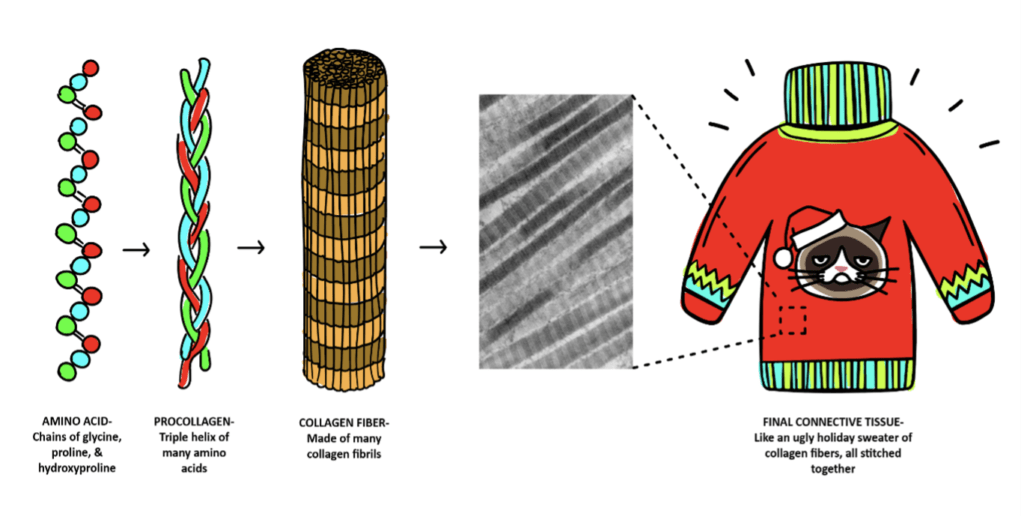
Collagen is a large protein consisting of amino acids (the building blocks of proteins) bound together to form a triple-helical structure. This triple helix forms from many amino acids, but from an especially large quantity of the amino acids glycine and proline (and hydroxyproline which comes from proline). These amino acids form the triple helix, which together forms procollagen, fibrils, fibers, and ultimately the large collagen protein itself, of which there are different “types” based on which other amino acids decide to join the party.
You can think of collagen formation as the creation of a piece of clothing. More accurately, think of it as the creation of an entire wardrobe- since there are so many different types that provide different functions to so many tissues. The production of collagen starts with the preformed version of collagen, procollagen, which for the sake of this analogy is like the raw unwoven cotton material (we’re keeping this analogy wardrobe vegan-friendly). Procollagen molecules start to bind together into collagen fibrils which are like a single cotton thread. These fibrils then weave together to form structured collagen fibers, which are like thicker strands, which then get stitched together to form the final piece of fabric to make a shirt or pants or that ugly holiday sweater (the various connective tissues needed throughout the body). To make sure this production line runs smoothly and can generate these various items, it needs vitamin C, amino acids, cofactor nutrients, and enzymes. The problem is that, as we age, the collagen factory starts to slow down its production each year starting in your 30s. It’s okay. Breathe. We can accept aging with dignity and grace. My eye may or may not be twitching as I write this.
Dozens of collagen “types” have been identified in the human body and every type has a different amino acid composition with a specific role. Among the many types of collagen that have been identified, type I, II, and III are the most prevalent; these are responsible for tissue strength, elasticity, and water retention capacity. Type I provides tensile strength for tendons, bones, skin, and fascia in organs, which means it can be stretched and withstand strong forces without breaking. Gram for gram, type I collagen is stronger than steel! So, when you say you have buns of steel, that isn’t an exaggeration *wink*. Type II collagen is the main collagen in cartilage. This collagen type is essential for the synthesis and reconstruction of connective tissue and helps with joint flexibility, which is why studies looking at joint pain tend to use type II for supplementation. It is thought that type II collagen supplementation acts on the autoimmunity of the body’s own collagen in the joints, which may result in less inflammation in people with arthritis. Type III collagen is an important component of the interstitial tissue of the lungs, liver, skin, spleen, and blood vessels.
Collagen Is a Big Ol’ Protein
Since collagen types I, II, and III are the most common in the body, it makes sense that you will see these three types listed most on supplement tubs. You will also see “hydrolyzed collagen” or “collagen peptides” which are just smaller, broken-down chains of collagen, usually a chain of two or three amino acids (di- and tripeptides). A selling point for collagen supplements is that they are easily absorbed and utilized by the body. This is a bit deceiving.
It is true that peptides with shorter chains, and di- and tripeptides, are easily absorbed through the gastrointestinal tract. In fact, they are absorbed more quickly than individual amino acids are, since single amino acids must compete for transport through the small intestine’s lining- This is why supplementing with individual amino acids is a bad idea since it will create a “traffic jam” of sorts and block each other’s absorption. So, yes, a smaller chain, like a collagen peptide, can theoretically absorb efficiently into the intestinal cells. However, most small peptides are then broken down into amino acids prior to being absorbed into the bloodstream. Very few short-chain peptides enter the bloodstream intact. Large proteins like collagen, or smaller proteins, like collagen peptides, will break down into the basic building blocks of all proteins: Amino acids.
Amino acids are constantly being broken up, reordered, and refolded into whatever the body needs to build at the time- like for repairing and building tissue, muscle, DNA and RNA, enzymes, hormones, and for producing energy to some extent. Our bodies are much smarter than we are (no offense) and will prioritize the synthesis of different proteins based on what we need the most. In other words, it’s impossible to know where collagen peptides or individual amino acids will be used in the body, or if they will be used to produce collagen at all, since the peptides coming from collagen may just as well be used to make other proteins. Your body can not tell whether it gets peptides and amino acids from collagen directly, or from any other source. Once collagen is broken down and absorbed, its amino acids become part of the amino acid pool and made into whatever the body needs- Your body doesn’t care whether you want that glowing skin or not. Womp womp.
So, we do not need to eat collagen to make collagen, since our bodies build collagen from available amino acids, just like it does with lots of proteins. But we do need to make sure the nutrients playing a role in collagen synthesis are present to complete its formation, including getting adequate protein for our individual needs.
Vegan Collagen?
Now that we’ve gone over what collagen actually is, let’s pop this bubble: Collagen is not vegan. Fish, pigs, cows, and chickens are all used to source collagen supplements. Further, all the collagen supplement research used collagen or gelatin sourced from animals.
You will find plenty of products if you do a search for “vegan collagen.” But be aware that these do not actually contain collagen because collagen is an animal-based protein. Notice how vegan collagen products will use the term collagen “support,” “builder,” or “booster.” That is because these are blends of different amino acids, vitamins (especially vitamin C), minerals, extracts, and/or herbal concoctions. The idea with these supplements is that the combination of their ingredients will help your body bump up collagen production with “pro-collagen nutrients,” but you will not be ingesting collagen directly with these. Some plants are purported to have the ability to promote the synthesis of procollagen and type I collagen, like camelia, amla, ginseng root, and cinnamon, so you may see such herbs in vegan collagen-boosting supplements.
It is up to you whether you want to add these boosters to your routine, but we can get all the nutrients we need to make collagen, either from our body synthesizing them on its own or from diet alone. To summarize: When we are talking about supplementing with collagen, these are the ways to do so: 1. Consume direct sources of collagen or collagen peptides which come from animal connective tissues (not vegan) 2. Consume supplements with the nutrients that boost the body’s natural production of collagen (these do not contain collagen) OR 3. Get all the nutrients your body needs to form its own collagen with food.
I’ve seen talk around the internets that biotech companies are working toward growing collagen in a lab with a fermentation process, rather than sourcing it from animals. I have not found these products on the market yet, or more information about what this would mean for vegan collagen supplements, but maybe the collagen market of the future is a vegan one. For now, however, all research involves animal-based collagen protein. What does the literature say about collagen supplementation?
Collagen Research
Athletics: The claim in sports nutrition is that collagen supplements can increase lean muscle gain, decrease recovery time, rebuild damaged joints, and improve overall cardiovascular performance. In 2018, sports nutrition researchers published a dietary supplement review for the International Olympic Committee (IOC). They state that potential benefits of collagen supplementation include: “Increased collagen production, thickened cartilage, [and] decreased joint pain.” They go on to conclude that “gelatin and collagen supplements appear to be low risk. Few data [is] available, but increased collagen production and decreased pain seem possible. Functional benefits, recovery from injury, and effects in elite athletes are not known.” I’m getting a lot of shrug vibes. Some of the studies included in their review found statistically significant improvements in joint pain, increase in collagen synthesis, and increase in cartilage thickness in individuals with arthritis. Specifically, a study of active men found an increase in fat-free mass with no change in muscle fiber, where the authors suggest that the increase in fat-free mass could be due to a growth in connective tissue rather than muscle growth. The consensus overall was that a few studies showed a small potential benefit from collagen or gelatin supplementation, but overall, there is not enough evidence to make a strong conclusion that supplementation was helpful. The studies included in the IOC review supplemented with either collagen or gelatin with the addition of vitamin C. As such, if you were to supplement with collagen, the IOC recommended dose is 5-15g of gelatin or 10g/day of collagen with 50mg of vitamin C. A review from 2020 echoed conclusions made by the IOC review, that dietary collagen does not appear to stimulate muscle protein synthesis. However, there is evidence that it may promote favorable body composition and help with connective tissue remodeling when combined with exercise. They go on to say collagen protein in combination with other animal or plant-based protein sources may help to maintain or improve connective tissue and may provide some benefit to bone health. It makes sense that collagen would need to be combined with other protein sources to see a benefit- Collagen is low in leucine, a particularly important amino acid for initiating muscle protein synthesis for muscle growth, so it doesn’t necessarily make an ideal muscle-building protein source on its own.
COACHES NOTE! Hi, Dani here. After reading through this incredible article, I had questions about these athletic studies. Were these folks just taking in additional protein in the form of collagen? If so, couldn’t that alone account for the potential improvement in connective tissue remodeling. After Natalie and I looked at the studies closer, of the four studies references, only ONE of them controlled for nutrition. The other three did not control for any other variables which is very interesting and leads me to further believe that it’s possible that any improvements could be chalked up to an overall increase in protein. Take it away Natalie…
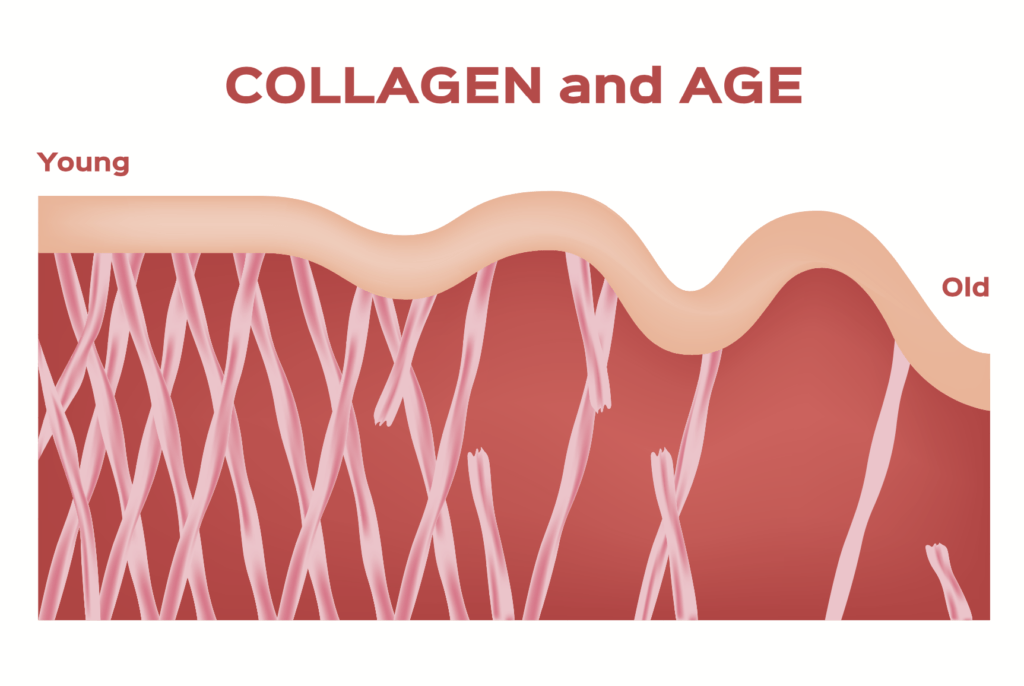
Beauty: Collagen is the most abundant component of the extracellular matrix of the skin, constituting 75% of skin’s dry weight. As we age, collagen becomes fragmented, kind of like a fraying rope, and we also produce less of it; we lose approximately 1% of overall collagen content in the skin each year! This results in less-firm, wrinkled skin. SO FUN (I’m actually crying a little). Specifically, this is likely due to the loss of collagen type I. Supplemental collagen is thought to increase the synthesis of type I collagen and hyaluronic acid (another important component of skin). A 2019 review of randomized control trials on collagen supplementation published in the Journal of Drugs in Dermatology found that doses ranging from 2.5 to 10 grams of collagen hydrolysate or collagen peptides per day may increase skin elasticity, skin hydration, and dermal collagen density. However, this review only included a few studies with small study groups over the span of a few months.
Bottom Line
Altogether, there is preliminary evidence that supplementation with either gelatin or collagen, combined with vitamin C, may have a small, negligible to modest positive effect for individuals with joint pain or issues with connective tissue. However, there is not enough evidence to make a recommendation. Additionally, a lot of studies are small, short-term, based on mice, or are industry-funded. It also seems like a lot of studies combine both objective measures (like skin biopsies) along with subjective data (like visual scales and patients’ self-reporting). Until we have more data, there is not much of a conclusive answer. The research overall seems to be in its beginning stages, and we need much more long-term data in general regarding the potential benefits.
Collagen, like any protein, gets broken down during digestion into small peptides and amino acids, which are used to make new proteins- collagen or anything else, depending on what your body needs. Unless your diet is protein deficient (unlikely if you are consuming adequate calories), loading up on more amino acids by taking collagen supplements is unlikely to make much of a difference. And, in case you missed it above, collagen (and gelatin) is not vegan; direct supplementation is off the table if you want to stick with plant-based options.
Taking collagen boosting supplements (which are vegan) is a matter of preference if you feel your diet is lacking and you don’t mind spending the extra cash. But theoretically, a well-balanced vegan diet, adequate in protein, with sufficient intake of vitamin C-rich plant foods, and the nutrients needed for cofactor and enzyme production, should be enough for collagen synthesis. It is worth mentioning certain lifestyle choices can damage collagen or slow down its production. Stress, smoking, sun damage, and high sugar intake can all increase oxidative stress, chronic inflammation, and the breakdown of collagen fibers.
While there does not seem to be harmful effects reported in the small body of literature of collagen supplementation, be aware that large doses of hydroxyproline can increase your risk of kidney stones if you are predisposed to them, since hydroxyproline breaks down into oxalates. If you feel your connective tissues need extra recovery, maybe that would be a time to consider supplementing. However, it could be more beneficial to see if there are other areas in your training that can be modified to assist in recovery, like taking a de-load week, for example.
It would be interesting to see how effective collagen is in the context of a healthy person who is already eating a diet that has plenty of vitamin C and plenty of the constituent amino acids that are necessary to synthesize collagen. Would the potential impact of these supplements be less significant with people that ensure their diet is made up of collagen-making goods? It’s hard to say. But the short answer is: Vegans can consume and synthesize all the necessary amino acids and nutrients for collagen production with a well-balanced diet. So my fine lines, my bum knees, and I will be doing just that. Which leads us to…
Nutrients For Collagen Synthesis:
Surprise, surprise, the dietitian recommends focusing on food first before considering supplements! If you want to ensure you are getting the nutrients necessary to properly synthesize collagen in your diet, focus on getting the protein, vitamins, and minerals that form collagen’s structure and act as cofactors for enzymes. Here are the collagen-building nutrients your body needs and where to find them:
Amino Acids: Having protein from a variety of plant foods throughout the day is enough to make sure your body will have all the amino acids you need for building proteins. If you are getting adequate protein from a variety of sources, you should be getting the necessary amino acids to synthesize collagen. The Recommended Dietary Allowance (RDA) for protein in adults is 0.8 grams of protein per kilogram of body weight, but you may need more depending on your current needs and goals.
Glycine is the primary amino acid in collagen, making up one-third of its amino acid structure. Glycine is not an essential amino acid, which means you do not need to get it from food. However, food sources of glycine include soy, beans, peanuts, sesame and sunflower seeds, spirulina, watercress, leeks, spinach, amaranth, and wheat germ.
After glycine, the next largest amino acid component in collagen is formed by proline, and hydroxyproline, which together comprise approximately 23% of the collagen molecule. Hydroxyproline, found in few proteins other than collagen, allows for the sharp twisting shape of the collagen helix. Proline is considered a “conditionally essential amino acid” which means our bodies can make it on their own but may need additional reinforcement during high-stress times, like in wound healing. Food sources of proline include asparagus, mushrooms, beans, buckwheat, cabbage, chives, cucumbers, garbanzo beans, peanuts, soy, watercress, and mustard seed. Arginine, another amino acid, is involved in collagen formation by being a precursor to proline.
Another amino acid of note is lysine, which forms hydroxylysine and helps shape collagen and protects it from enzymatic breakdown. Like hydroxyproline, hydroxylysine is also found in few proteins other than collagen. Lysine is an essential amino acid, meaning your body can not make it on its own, so you must get it from food. The RDA for lysine is 38 mg/kg/day. Sources of lysine include peas, beans, and lentils, and legume-based protein powders, tempeh, tofu, edamame, and other soy foods like meat analogs, cashews, pistachios, pumpkin seeds, buckwheat, and quinoa.
Vitamin C: Other than amino acids, vitamin C is perhaps the most important nutrient involved in collagen synthesis. Vitamin C is well-known for its role in promoting connective tissue healing after injury and this is because of its essential part in collagen synthesis. One of the hallmark associations of vitamin C deficiency is scurvy, which has symptoms that include the breakdown of skin, gums, and other collagenous tissues. Yikes! Specifically, vitamin C is a necessary cofactor (this is like a helper catalyst to an enzyme…which is a catalyst to metabolic pathways…double catalyst!) for the enzymes that hydroxylate (adds a hydrogen and an oxygen to) proline and lysine so that they can form collagen’s triple helix shape. It also promotes collagen gene expression. Women need 75 mg/day, and men need 90 mg/day. Luckily, vitamin C is found in abundance in the plant world. Food sources include dark leafy greens, cruciferous veggies like broccoli, kale, Brussels sprouts, and cauliflower, tomatoes, red bell pepper, citrus fruits, kiwi, mango, papaya, strawberries, and pineapple.
Zinc: Zinc is a cofactor that helps activate collagen production. Especially during wound healing, zinc activates an enzyme called collagenase that allows cells to remodel collagen. Men need 11 mg/day and women need 8 mg/day. Food sources include beans like chickpeas, cashews, almonds, pumpkin seeds, hemp seeds, sesame seeds, wheat germ, and cocoa.
Manganese: Manganese acts as a cofactor for the enzyme prolidase, which is the enzyme responsible for proline formation, one of the important amino acids in collagen’s structure. Women need 1.8 mg/day and men need 2.3 mg/day. Foods sources include beans like pinto, lima, and navy beans, hazelnuts, pecans, pumpkin seeds, tofu, and whole grains like brown rice.
Copper: Copper supports the formation of fibroblasts (cells that synthesizes collagen), stimulates collagen production (especially types I, II and V), and helps to build collagen fibril structures. Lysyl oxidase is a copper-dependent enzyme that bonds collagen fibers with other supportive fibers to create a strong framework. Adults need 900 micrograms of copper daily. Food sources include sunflower seeds, lentils, almonds, apricots, chocolate, mushrooms, blackstrap molasses, kale, sesame seeds, cashews, and prunes.
Sulfur: Sulfur is a trace mineral that helps synthesize and prevent the breakdown of collagen. A very small amount of sulfur comes from food, but most of it is derived from the sulfur-containing amino acids, cysteine, and methionine (which is an essential amino acid). Food sources of sulfur include garlic, onions, cruciferous vegetables like kale, cabbage, and broccoli. Food sources of methionine include tofu, soy milk, edamame, nuts like hemp and pumpkin seeds, beans, and whole grains like quinoa.
Sources:
- Albaugh VL, Mukherjee K, Barbul A. Proline Precursors, and Collagen Synthesis: Biochemical Challenges of Nutrient Supplementation and Wound Healing. J Nutr. 2017;147(11):2011-2017. doi:10.3945/jn.117.256404
- Asserin, J., Lati, E., Shioya, T. and Prawitt, J. (2015), The effect of oral collagen peptide supplementation on skin moisture and the dermal collagen network: evidence from an ex vivo model and randomized, placebo‐controlled clinical trials. J Cosmet Dermatol, 14: 291-301. https://doi.org/10.1111/jocd.12174
- Bagchi D, Misner B, Bagchi M, Kothari SC, Downs BW, Fafard RD, Preuss HG. Effects of orally administered undenatured type II collagen against arthritic inflammatory diseases: a mechanistic exploration. Int J Clin Pharmacol Res. 2002;22(3-4):101-10. PMID: 12837047.
- Barbul A. Proline precursors to sustain Mammalian collagen synthesis. J Nutr. 2008 Oct;138(10):2021S-2024S. doi: 10.1093/jn/138.10.2021S. PMID: 18806118.
- Binic I, Lazarevic V, Ljubenovic M, Mojsa J, Sokolovic D. Skin aging: natural weapons and strategies. Evid Based Complement Alternat Med. 2013;2013:827248. doi:10.1155/2013/827248
- Bolke L, Schlippe G, Gerß J, Voss W. A Collagen Supplement Improves Skin Hydration, Elasticity, Roughness, and Density: Results of a Randomized, Placebo-Controlled, Blind Study. Nutrients. 2019; 11(10):2494.
- Borkow G. Using Copper to Improve the Well-Being of the Skin. Curr Chem Biol. 2014;8(2):89-102. doi:10.2174/2212796809666150227223857
- Choi FD, Sung CT, Juhasz ML, Mesinkovsk NA. Oral Collagen Supplementation: A Systematic Review of Dermatological Applications. J Drugs Dermatol. 2019 Jan 1;18(1):9-16. PMID: 30681787.
- Clark KL, Sebastianelli W, Flechsenhar KR, Aukermann DF, Meza F, Millard RL, et al. 24-Week study on the use of collagen hydrolysate as a dietary supplement in athletes with activity-related joint pain. Curr Med Res Opin. 2008 May;24(5):1485–96.
- Deane et al. Animal, Plant, Collagen and Blended Dietary Proteins: Effects on Musculoskeletal Outcomes. Nutrients 2020, 12(9), 2670; https://doi.org/10.3390/nu12092670
- DePhillipo NN, Aman ZS, Kennedy MI, Begley JP, Moatshe G, LaPrade RF. Efficacy of Vitamin C Supplementation on Collagen Synthesis and Oxidative Stress After Musculoskeletal Injuries: A Systematic Review. Orthop J Sports Med. 2018;6(10):2325967118804544. Published 2018 Oct 25. doi:10.1177/2325967118804544
- Ganceviciene R, Liakou AI, Theodoridis A, Makrantonaki E, Zouboulis CC. Skin anti-aging strategies. Dermatoendocrinol. 2012;4(3):308-319. doi:10.4161/derm.22804
- Hashim, P., Mohd Ridzwan, M. S., Bakar, J. and Mat Hashim (2015), D. Collagen in food and beverage industries. International Food Research Journal 22(1): 1 – 8 (2015). http://psasir.upm.edu.my/id/eprint/36110/1/(1).pdf
- Kirmse M, Oertzen-Hagemann V, de Marées M, Bloch W, Platen P. Prolonged Collagen Peptide Supplementation and Resistance Exercise Training Affects Body Composition in Recreationally Active Men. Nutrients. 2019 May;11(5).
- Knight J, Jiang J, Assimos D, Holmes R. Hydroxyproline ingestion and urinary oxalate and glycolate excretion. Kidney Int. 2006 Dec;70(11):1929–34.
- Kumar, S., Sugihara, F., Suzuki, K., Inoue, N. and Venkateswarathirukumara, S. (2015), A double‐blind, placebo‐controlled, randomized, clinical study on the effectiveness of collagen peptide on osteoarthritis. J. Sci. Food Agric., 95: 702-707. https://doi.org/10.1002/jsfa.6752
- Lindsay Allen, Andrew Prentice, Encyclopedia of Human Nutrition. Elsevier, Jul 20, 2005. Pages (https://books.google.com/books?id=DHtERWm0mrcC&printsec=frontcover#v=onepage&q&f=false)
- Lis DM, Baar K. Effects of Different Vitamin C-Enriched Collagen Derivatives on Collagen Synthesis. Int J Sport Nutr Exerc Metab. 2019 Sep 1;29(5):526–31.
- Lodish H, Berk A, Zipursky SL, et al. Molecular Cell Biology. 4th edition. New York: W. H. Freeman; 2000. Section 22.3, Collagen: The Fibrous Proteins of the Matrix. Available from: https://www.ncbi.nlm.nih.gov/books/NBK21582/
- Lugo, J.P., Saiyed, Z.M. & Lane, N.E. Efficacy and tolerability of an undenatured type II collagen supplement in modulating knee osteoarthritis symptoms: a multicenter randomized, double-blind, placebo-controlled study. Nutr J 15, 14 (2015). https://doi.org/10.1186/s12937-016-0130-8
- Maughan RJ, Burke LM, Dvorak J, Larson-Meyer DE, Peeling P, Phillips SM, et al. IOC Consensus Statement: Dietary Supplements and the High-Performance Athlete. Int J Sport Nutr Exerc Metab. 2018 Mar 1;28(2):104–25.
- McAlindon TE, Nuite M, Krishnan N, Ruthazer R, Price LL, Burstein D, et al. Change in knee osteoarthritis cartilage detected by delayed gadolinium-enhanced magnetic resonance imaging following treatment with collagen hydrolysate: a pilot randomized controlled trial. Osteoarthritis Cartilage. 2011 Apr;19(4):399–405.
- National Nutrient Database for Standard Reference. U.S. Department of Agriculture. Retrieved 2021-01.
- Praet SFE, Purdam CR, Welvaert M, Vlahovich N, Lovell G, Burke LM, Gaida JE, Manzanero S, Hughes D, Waddington G. Oral Supplementation of Specific Collagen Peptides Combined with Calf-Strengthening Exercises Enhances Function and Reduces Pain in Achilles Tendinopathy Patients. Nutrients. 2019 Jan 2;11(1):76. doi: 10.3390/nu11010076. PMID: 30609761; PMCID: PMC6356409.
- Proksch E, Segger D, Degwert J, Schunck M, Zague V, Oesser S. Oral supplementation of specific collagen peptides has beneficial effects on human skin physiology: a double-blind, placebo-controlled study. Skin Pharmacol Physiol. 2014;27(1):47-55. doi: 10.1159/000351376. Epub 2013 Aug 14. PMID: 23949208.
- Pullar JM, Carr AC, Vissers MCM. The Roles of Vitamin C in Skin Health. Nutrients. 2017;9(8):866. Published 2017 Aug 12. doi:10.3390/nu9080866Woo T, Lau L, Cheng N, Chan P, Tan K, et al. (2017) Efficacy of Oral Collagen in Joint Pain – Osteoarthritis and Rheumatoid Arthritis. J Arthritis 6: 233. doi:10.4172/2167-7921.1000233
- Rawson, E. S., Miles, M. P., & Larson-Meyer, D. (2018). Dietary Supplements for Health, Adaptation, and Recovery in Athletes, International Journal of Sports Nutrition and Exercise Metabolism, 28(2), 188-199. Retrieved Jan 29, 2021, from https://journals.humankinetics.com/view/journals/ijsnem/28/2/article-p188.xml
- Shaw G, Lee-Barthel A, Ross ML, Wang B, Baar K. Vitamin C-enriched gelatin supplementation before intermittent activity augments collagen synthesis. Am J Clin Nutr. 2017;105(1):136–43.
- SJ Padayatty, M Levine. Vitamin C: the known and the unknown and Goldilocks. Oral Diseases (2016) 22, 463–493 doi:10.1111/odi.1244
- William van B. Robertson, Metabolism of Collagen in Mammalian Tissues. Biophysical Journal. Volume 4, Issue 1, Part 2, 1964. Pages 93-106. ISSN 0006-3495. https://doi.org/10.1016/S0006-3495(64)86930-7
- Yamauchi M, Sricholpech M. Lysine post-translational modifications of collagen. Essays Biochem. 2012;52:113-133. doi:10.1042/bse0520113
- Zdzieblik, D., Oesser, S., Baumstark, M., Gollhofer, A., & König, D. (2015). Collagen peptide supplementation in combination with resistance training improves body composition and increases muscle strength in elderly sarcopenic men: A randomized controlled trial. British Journal of Nutrition, 114(8), 1237-1245. doi:10.1017/S0007114515002810